Showroom
The mentioned Generic Medicines find usage in various treatments such as nausea, cough, headache, neck pain, cold, stomach ache, vomiting, nose bleeding, constipation, allergy and many more. This range also includes all the medicines required by a smoker to quit smoking. The effect of these medicines is same as that of branded ones but have lower costs.
Zop(zopiclone tablets), accuzon 2 g(ceftriaxone injection), tadora 20 mg tablets are some of the many pharmaceutical drugs one can find at our store. Our company trades in these drugs, supplying the same purchased from manufacturers to the market.
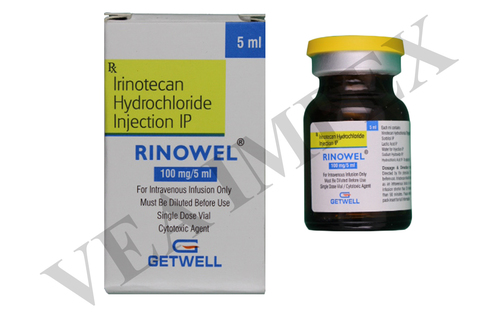
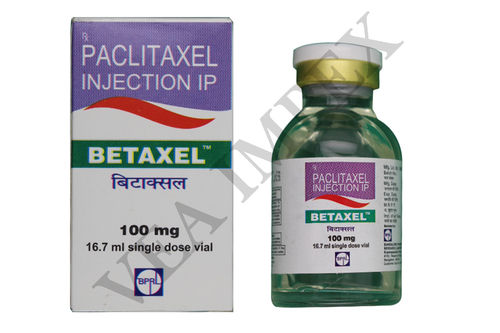
Our offered Anti Cancer Medicines prevent the growth of neoplasms by destroying the cancer cells. In addition, these also inhibit the development of additional blood supply that leads to the growth of cancer cells. These are not only used for treating different kind of cancers but in the conjunction with several therapies and surgeries.
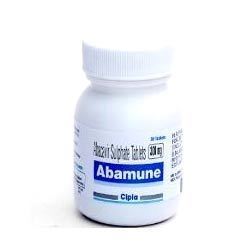
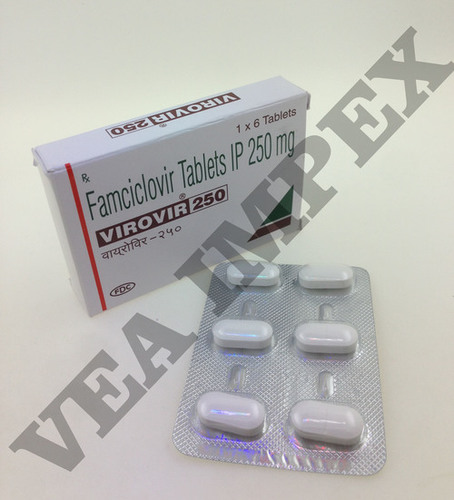
HIV AIDS Medicines helps in decreasing lethal virus known as Human immunodeficiency virus by strengthening the immune system. These medicines are also known as antiretroviral medicines and work by preventing the virus to make several copies of itself. This helps in providing strength to the immune system for its proper functioning by developing more amount of CD4 T cells.
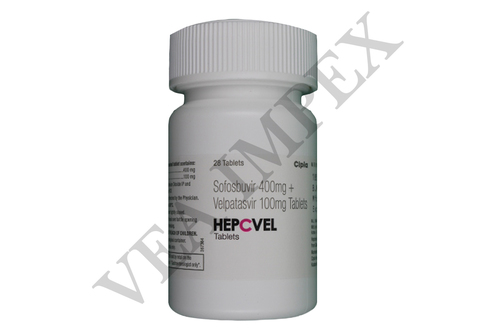
Hepatitis refers to the inflammation caused in the tissues of liver. Our offered Hepatitis Medicines are used for treating symptoms such as tiredness, nausea, fever, diarrhea and headache. The main ingredients used in these medicines are Peginterferon and ribavirin that help in preventing the growth of bacteria which causes hepatitis. Hepatitis A, Hepatitis B both can be treated by using this range of medicines.
All the herbal products offered under the category of Himalaya Tablets are made by HIMALAYA WELLNESS. These are suitable for people of all ages and are made using natural herbs. Concept of Ayurveda has been combined with latest science in these tablets.
IV Fluids - Intravenous Infusion are widely used to maintain a patient's hydration, electrolyte and blood sugar levels while undergoing surgical procedures. These fluids are also used in treatment of an infection using antibiotics. These fluids are help in management of pain using certain medications.
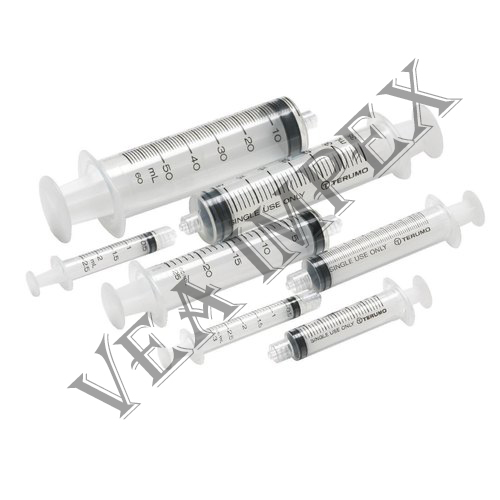
Surgical Products are provided by our company which act as a protective gear designed to safeguard the health of workers by minimizing the exposure to a biological agent. They are specialized clothing or equipment worn by an employee for protection against infectious materials.
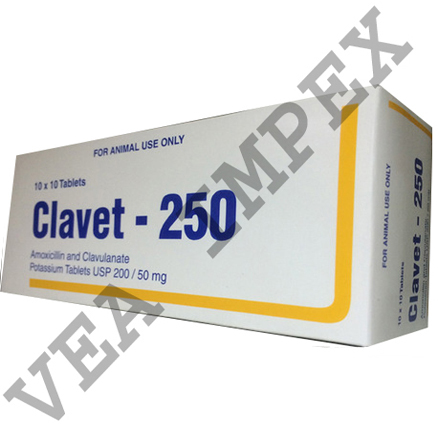
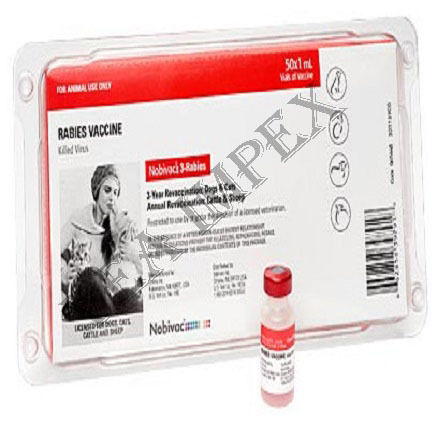
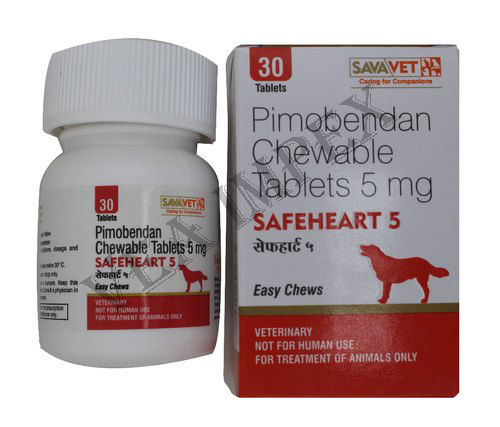
Veterinary Products & Medicine is the branch of medicine that deals with the prevention, control, diagnosis, and treatment of disease, disorder, and injury in animals. This medicine ensures a safe food supply for people by monitoring and maintaining the health of food-producing animals.
Mosquito Repellent is a blend of extracts from plants such as eucalyptus, lemongrass, neem among others, and many other aromatic oils. This is an excellent alternative to chemical-based repellents. It causes no respiratory problems and has no other side effects. This repellent is very effective and economical too.
COVID - 19 Medicine & Products are used for protecting you from the effect of virus, keeping you safe and secure as well as healthy too. These products are very effective and are recommended by doctors to keep people safe. They are made using premium grade materials and are easy to wear and discharge too.
Antiseptic and Disinfectant acts as a chemical agent which are applied to non-living objects in order to destroy bacteria, viruses, fungi, mould or mildews living on the objects. This offered agent is widely appreciated by our customers, in the market.
Pain Relief Spray is used to treat minor aches and pains of the muscles/joints. This spray works by causing the skin to feel cool and then warm. This spray tends to work best on joints that are close to the surface of the skin, such as the joints in the hands or knees.
Toothpaste is mainly used for promoting the oral hygiene. This paste is used for cleaning the teeth in a better way. This paste aids in removing dental plaque and food from the teeth. This plays a unique role in keeping your teeth clean and healthy. This paste help to prevent tooth decay and gum disease.
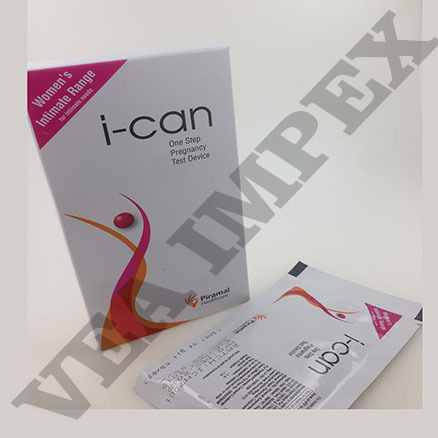
Pregnancy Test Kit is widely used by women to check and test their chances of being pregnant or not. This can be easily used at homes. This kit is easy to use, comfortable and affordable too. This kit can be used at any time and as per your convenience.
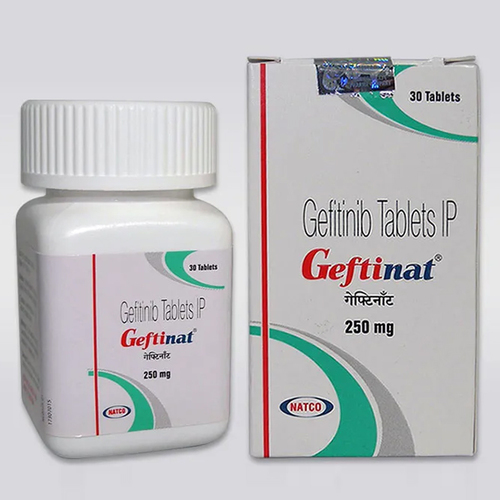
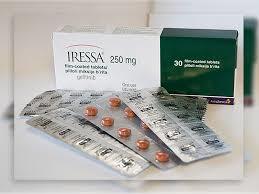
Lung Cancer Medicine is safe, effective, and possibly better than the current standard. This reduces the chances of recurrence, while there is always a chance that cancer will return. This medicine is very much liked and highly appreciated by people, in the market.
"We mainly deliver our products to the foreign countries."

 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese


































 Call Me Free
Call Me Free