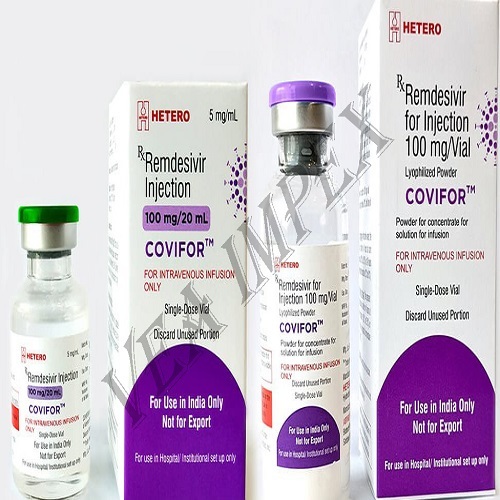
Remdesivir injection
Product Details:
- Salt Composition Remdesivir
- Origin of Medicine India
- Drug Type Injection
- Physical Form Tablets
- Function COVID-19
- Dosage As directed by the physician
- Suitable For Suitable For All
- Click to view more
X
Remdesivir injection Price And Quantity
- 5000-1000 Piece
Remdesivir injection Product Specifications
- Cool and Dry place
- COVID-19
- India
- Remdesivir
- Injection
- Tablets
- Suitable For All
- As directed by the physician
Remdesivir injection Trade Information
- Nhava Sheva
- 10000 Piece Per Week
- 7-15 Days
- Yes
- Sample costs shipping and taxes has to be paid by the buyer
- As per customer request
- Australia, North America, South America, Eastern Europe, Western Europe, Middle East, Africa, Asia, Central America
- All India
- WHO,GMP
Product Description
Uses
Remdesivir is an investigational drug being studied to treat coronavirus infection, also known as COVID-19. It has not been approved by the FDA for general use. However, the FDA is now allow ingremdesivir to be used in human studies and for emergency use in certain hospitalized patients. More information should be provided by the doctor from either the Informed Consent form for a study or the FDA Fact Sheet for Patient sand Parents/Caregivers for emergency use.
How to use Remdesivir 100 Mg Intravenous Powder For Solution (EUA)
Remdesivir is given by injection into a vein usually once daily for 5 to 10 days. It is infused over a period ranging from 30 minutes to 2 hours.
Effective COVID-19 Treatment
Remdesivir Injection is valued for its role in addressing moderate to severe COVID-19 symptoms. It hampers the replication of the virus within the body, potentially reducing the severity and duration of the illness when administered early in the course of infection. Healthcare providers trust Remdesivir for its efficacy and safety in professional settings.
Safe Handling and Storage Guidelines
To ensure the quality and effectiveness of Remdesivir Injection, it is essential to store it in a cool and dry environment. This precaution maintains the medications stability and potency. Proper storage is a key element in preserving the medication before administration and during distribution to healthcare facilities.
FAQs of Remdesivir injection:
Q: How should Remdesivir Injection be used for treating COVID-19?
A: Remdesivir Injection must be administered under the supervision of a qualified physician specifically for treating COVID-19. The exact dosage and duration will be determined by your healthcare provider based on your individual condition.Q: What is the benefit of using Remdesivir Injection?
A: Remdesivir Injection works by inhibiting the replication of the virus that causes COVID-19, thereby helping reduce the severity and recovery time of the infection when used as recommended.Q: When is it appropriate to start Remdesivir Injection for COVID-19?
A: Remdesivir should ideally be started early in the course of the illness for maximum efficacy. The timing and necessity will be decided by the attending physician based on the patients clinical presentation.Q: Where should Remdesivir Injection be stored to maintain its effectiveness?
A: Remdesivir Injection should be stored in a cool and dry place, away from direct sunlight and moisture to preserve its stability and medicinal properties.Q: What is the process for obtaining Remdesivir Injection as an exporter, supplier, or wholesaler?
A: Remdesivir Injection is available through authorized pharmaceutical exporters, suppliers, traders, and wholesalers in India. Appropriate certifications and compliance with regulatory protocols are necessary for procurement and distribution.Q: Is Remdesivir Injection suitable for all patients?
A: Remdesivir Injection is suitable for all individuals prescribed it by a healthcare professional. However, the physician will assess suitability based on specific patient health conditions and requirements.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
Other Products in 'COVID - 19 Medicine & Products' category
"We mainly deliver our products to the foreign countries."