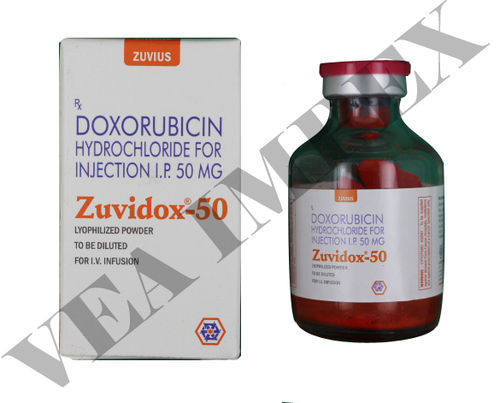

Zuvidox 50mg (Doxorubicin Hydrochloride Injection)
Product Details:
- Indication Cancer treatment
- Dosage Form Injection
- Origin India
- Salt Composition Doxorubicin Hydrochloride
- Feature Other
- Application Other
- Fermentation Smell Normal Smell
- Click to view more
Zuvidox 50mg (Doxorubicin Hydrochloride Injection) Price And Quantity
- 5000-1000 Piece
- 950 INR/Piece
Zuvidox 50mg (Doxorubicin Hydrochloride Injection) Product Specifications
- Other
- Other
- 12 Months
- Doxorubicin Hydrochloride
- Injection
- Store below 30C
- Cancer treatment
- India
- Normal Smell
Zuvidox 50mg (Doxorubicin Hydrochloride Injection) Trade Information
- 10000 Piece Per Week
- 7-15 Days
- Middle East, Africa, South America, Western Europe, Asia, Central America, Eastern Europe, North America, Australia
- All India
Product Description
Doxorubicin Hydrochloride Injection, also known as Zuvidox 50mg, is a cytotoxic chemotherapy drug used to treat various malignancies. Doxorubicin causes the fast division of cancer cells to die by preventing DNA replication. It is recommended for the treatment of solid tumours, including lung cancer, breast cancer, and ovarian cancer. A medical expert administers the injection intravenously. Hair loss, nausea, vomiting, and reduced blood cell counts are examples of typical adverse effects. Zuvidox 50mg has demonstrated effectiveness in treating certain tumours, perhaps providing advantages and bettering patient outcomes. During treatment, close medical care is required.
Zuvidox 50mg (Doxorubicin Hydrochloride Injection) Features Include:
1. Doxorubicin Hydrochloride, a strong cytotoxic chemotherapy drug used in cancer treatment, is the injection's active ingredient.
2. Various cancers, including breast cancer, ovarian cancer, lung cancer, and other solid tumours, are indicated for the treatment with Zuvidox 50mg.
3. Using an intravenous injection ensures that the drug is delivered quickly to the cancer cells it is intended to treat.
4. Accurate Treatment Planning is Made Possible by the Standardised and Precise Dosage of Doxorubicin Provided by Zuvidox 50mg.
5. Customised Treatment: Based on the patient's unique cancer type, stage, and general health state, the dosage and frequency of administration are adjusted.
Zuvidox 50mg (Doxorubicin Hydrochloride Injection) Benefits Include:
1. Doxorubicin has cytotoxic effects by preventing DNA replication in cancer cells that divide quickly, which causes the cells to die.
2. Zuvidox 50mg has shown effectiveness in controlling breast cancer, ovarian cancer, lung cancer, and other solid tumours, perhaps providing advantages and better results for people with these particular cancers.
3. Doxorubicin therapy may result in tumour stabilisation or decrease, which aids in the regulation of cancer growth.
4. Doxorubicin is frequently administered as adjuvant therapy after surgery or radiation to lower the chance of cancer recurrence.
5. Neoadjuvant Therapy: To reduce tumour size prior to surgery or other treatments, doxorubicin may occasionally be utilised.
6. Combining Doxorubicin with Other Therapies: To improve overall cancer management, doxorubicin may be combined with other chemotherapeutic drugs or therapies.
7. Potential for Longer Survival: Patients with some forms of cancer may see higher survival rates after receiving therapy with Zuvidox 50mg.
8. Doxorubicin can help people live better lives by reducing the signs and symptoms of their cancer.
9. Remission of the Illness: In a few instances, doxorubicin therapy may result in a partial or total remission of cancer.
Individual responses to Doxorubicin Hydrochloride Injection, Zuvidox 50mg, may differ, and strict medical supervision and monitoring are required to manage any potential side effects and optimise treatment outcomes. Throughout the course of treatment, patients should speak with their medical specialists for personalised counsel and direction.
Zuvidox 50mg (Doxycycline Hydrochloride Injection) Uses Include:
Several kinds of cancer are treated with Zuvidox 50mg, including:
1. Breast cancer: Both early and advanced stages of breast cancer are managed using it.
2. Ovarian Cancer: The medication Zuvidox 50mg is used to treat ovarian cancer.
3. Small cell lung carcinoma is one type of lung cancer for which it is suggested.
4. Soft Tissue Sarcoma: Treatment for soft tissue sarcomas involves the use of Zuvidox 50mg.
5. Multiple Myeloma: It may be used to treat the blood malignancy multiple myeloma.
6. Other Solid Tumours: A number of other solid tumours may be treated with the injection.
Zuvidox 50mg (Doxorubicin Hydrochloride Injection) Side Effects Include:
The adverse effects of Zuvidox 50 mg can take many different forms, and each person will experience them differently. Several frequent adverse effects include:
1. Vomiting and Nausea: Gastrointestinal side effects are frequently accompanied by nausea and vomiting.
2. Hair Loss: Zuvidox 50mg may cause temporary hair loss or thinning (alopecia).
3. Doxorubicin can lower the number of white blood cells, red blood cells, and platelets, which can raise the risk of infection, anaemia, and bleeding.
4. Weakness and weariness are possible side effects for patients.
5. Mouth Ulcers: Some people may experience mouth ulcers or sores.
6. Skin Reactions: Itching, rashes, and other skin-related problems could happen.
7. Doxorubicin can cause cardiac toxicity, thus patients may need to be closely watched for any indications of issues with their hearts.
8. Doxorubicin has the potential to alter the function of the liver and kidneys.
9. Fever: A side effect that some patients may experience is fever.
10. Diarrhoea: Some people may experience gastrointestinal side effects, such as diarrhoea.
Patients must notify their healthcare providers right once if any side effects are worrisome or persistent. In order to ensure the best possible management of cancer and address any potential side effects that may emerge, patients should also follow their prescribed dosages and undertake routine blood counts and heart function monitoring. In order to maximise therapy results and properly handle any adverse reactions, close medical care is required.
FAQs of Zuvidox 50mg (Doxorubicin Hydrochloride Injection):
Q: What is the dosage of Zuvidox 50mg (Doxorubicin Hydrochloride Injection)?
A: The dosage of Zuvidox 50mg (Doxorubicin Hydrochloride Injection) is 50 mg.Q: What is the storage instruction for Zuvidox 50mg (Doxorubicin Hydrochloride Injection)?
A: The storage instruction for Zuvidox 50mg (Doxorubicin Hydrochloride Injection) is to store below 30C.Q: What is the function of Zuvidox 50mg (Doxorubicin Hydrochloride Injection)?
A: The function of Zuvidox 50mg (Doxorubicin Hydrochloride Injection) is anti-cancer.Q: What is the physical form of Zuvidox 50mg (Doxorubicin Hydrochloride Injection)?
A: The physical form of Zuvidox 50mg (Doxorubicin Hydrochloride Injection) is liquid.Q: How should the dosage of Zuvidox 50mg (Doxorubicin Hydrochloride Injection) be administered?
A: The dosage of Zuvidox 50mg (Doxorubicin Hydrochloride Injection) should be administered as directed by the physician.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+